Wednesday, May 19, 2010
CCME to begin trading on the NASDAQ on June 3rd
FUJIAN, China--(BUSINESS WIRE)--China MediaExpress Holdings, Inc. (NYSE Amex: CCME) (“CME” or “Company”), China’s largest television advertising operator on inter-city and airport express buses, today announced that its common stock has been approved for listing on the NASDAQ Global Select Market.
Effective as of the close of trading on June 2, 2010, the Company’s common stock will cease trading on NYSE AMEX. Upon the opening of trading on June 3, 2010, the Company's shares will commence trading on NASDAQ Global Select Market, under the same ticker symbol "CCME".
Mr. Zheng Cheng, CME’s Founder and CEO, noted, “We believe that NASDAQ represents the best fit for our Company and our shareholders. NASDAQ's market maker structure should lead to increased liquidity, an expansion in our shareholder base and, ultimately, to increased shareholder value. In addition it will provide our investors with the best prices and the fastest execution. As the world's largest electronic stock market, NASDAQ promotes innovation and attracts leading growth companies from a diverse group of sectors. CME is one of the very few Chinese companies to qualify for the NASDAQ Global Select Market, which has the highest initial listing standards of any exchange in the world. Out of 136 China-based companies listed on NASDAQ, only 26 companies such as Baidu, CTrip.com International, Shanda Interactive Entertainment and NetEase.com, fulfill NASDAQ Global Select Market requirements. Within only seven months since CME became a public company in October 2009, we have qualified and are making the decision to move to the NASDAQ Global Select Market. This achievement is an important milestone for our Company and we look to the future with confidence.”
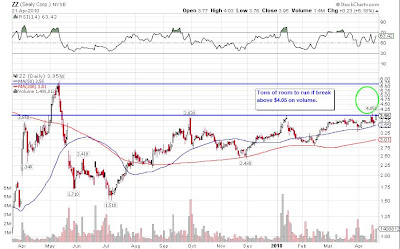
Long and Strong!
Monday, May 17, 2010
Thursday, May 13, 2010
Celsion's Phase III ThermoDox HEAT study Recommended for Continuation by Data Monitoring Committee
"COLUMBIA, Md., May 13 /PRNewswire-FirstCall/ -- Celsion Corporation (
The DMC for the HEAT study is comprised of an independent group of medical and scientific experts who are responsible for reviewing and evaluating patient safety and efficacy data. The DMC reviews study data at regular intervals with the charter to ensure patient safety and monitor the quality and overall conduct of the study. The study design and statistical plan for the Phase III ThermoDox trial also incorporates a pre-planned interim efficacy analysis by the DMC after patient enrollment is complete, with the intent to stop the study if there is overwhelming evidence of treatment benefit or a low probability of treatment success.
"We are pleased that the DMC has recommended continuation of the study based on its review of all of the available study data," stated Michael H. Tardugno, President and Chief Executive Officer of Celsion. "The DMC's affirmative review of the 12 Japanese patients enrolled to date is a positive step forward, as it confirms Celsion and Yakult's decision to initiate Phase III trials in Japan. The arrangement, to move directly into Phase III trials was based on previous evidence in primary liver cancer patients, may have shortened our timeline for approval in Japan by two years. With recent acceleration in patient recruitment, and the study now almost 60% enrolled, we are optimistic of a timely completion to enrollment."
Celsion's global Phase III ThermoDox study for primary liver cancer plans to enroll 600 patients and is being conducted under a FDA Special Protocol Assessment (SPA). The study is designed to evaluate the efficacy of ThermoDox in combination with radiofrequency ablation (RFA) when compared to patients who receive RFA alone as the control. The primary endpoint for the study is progression-free survival. Additional information on the Phase III ThermoDox clinical study may be found at www.clinicaltrials.gov."
LONG AND STRONG!! Accumulated more from last week.
Wednesday, May 12, 2010
Tuesday, May 11, 2010
Monday, May 10, 2010
MCGI Setup
Thursday, May 6, 2010
BROKEN TAPE
Wednesday, May 5, 2010
ARIA Trade
Tuesday, May 4, 2010
CRXX gap up play wins// overnight hold
Overnight hold update:
AKRX SCLN DVAX
Monday, May 3, 2010
Sunday, May 2, 2010
Friday, April 30, 2010
Thursday, April 29, 2010
Tuesday, April 27, 2010
Monday, April 26, 2010
Trades of the day
Friday, April 23, 2010
Thursday, April 22, 2010
ANTF setup update

Looks toppy here as volume continue to decrease while the stock keeps climbing higher.

Wednesday, April 21, 2010
DCTH Delcath Phase III Trial Results Exceed Primary Endpoint Expectations
NEW YORK, April 21 /PRNewswire-FirstCall/ -- Delcath Systems, Inc. (
James F. Pingpank, MD, FACS, Associate Professor of Surgery at the University of Pittsburgh School of Medicine and a lead Principal Investigator of the Phase III trial, will present full trial data on June 5th at the American Society of Clinical Oncology's (ASCO) 2010 Annual Meeting. The endpoints announced today are results of the independent core lab analysis, which will be submitted to support Delcath's New Drug Application (NDA) for its drug and proprietary treatment system to the
"We believe that these data support that the Delcath PHP System may provide a significantly better treatment option for patients suffering from melanoma metastases in the liver," said Eamonn P. Hobbs, President and CEO of Delcath. "With the treatment arm having a median hPFS of more than three-fold that of the control arm, we easily exceeded our expectations of clinical trial success. This is a major step forward in our plan to introduce what we believe is an effective treatment for patients who currently have very few viable options."
Tuesday, April 20, 2010
Monday, April 19, 2010
Overnight positions Chart setups April 19, 2010
Overnight positions $CHP $CUR $CYCC $KFS $PAL
PAL

KFS

CYCC

CUR

CHP