Friday, April 23, 2010
Thursday, April 22, 2010
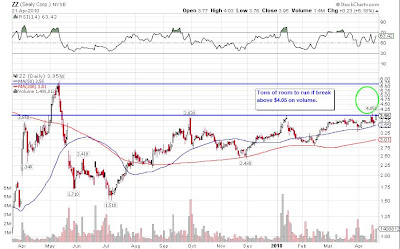
ANTF setup update

Looks toppy here as volume continue to decrease while the stock keeps climbing higher.

Wednesday, April 21, 2010
DCTH Delcath Phase III Trial Results Exceed Primary Endpoint Expectations
NEW YORK, April 21 /PRNewswire-FirstCall/ -- Delcath Systems, Inc. (
James F. Pingpank, MD, FACS, Associate Professor of Surgery at the University of Pittsburgh School of Medicine and a lead Principal Investigator of the Phase III trial, will present full trial data on June 5th at the American Society of Clinical Oncology's (ASCO) 2010 Annual Meeting. The endpoints announced today are results of the independent core lab analysis, which will be submitted to support Delcath's New Drug Application (NDA) for its drug and proprietary treatment system to the
"We believe that these data support that the Delcath PHP System may provide a significantly better treatment option for patients suffering from melanoma metastases in the liver," said Eamonn P. Hobbs, President and CEO of Delcath. "With the treatment arm having a median hPFS of more than three-fold that of the control arm, we easily exceeded our expectations of clinical trial success. This is a major step forward in our plan to introduce what we believe is an effective treatment for patients who currently have very few viable options."
Tuesday, April 20, 2010
Monday, April 19, 2010
Overnight positions Chart setups April 19, 2010
Overnight positions $CHP $CUR $CYCC $KFS $PAL
PAL

KFS

CYCC

CUR

CHP